Abstract
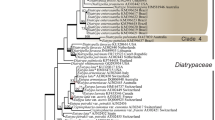
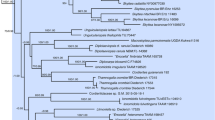
Botryotrichum iranicum (Chaetomiaceae) and Trematosphaeria magenta (Trematosphaeriaceae) are newly described from soils of Hyrcanian forests in the Mazandaran Province, Iran. Both species were distinguished from their closest known species based on multi-locus phylogenetic analysis and morphological characteristics. Botryotrichum iranicum is characterized by sympodially proliferating conidiophores, cylindrical and hyaline conidiogenous cells, hyaline, roughened, solitary conidia with distinctly visible hila and not forming asexual stage and sterile setae. Phylogenetic analyses of ITS, LSU sequences, and sequences part of the beta-tubulin gene support the recognition of this fungus as a new species. Trematosphaeria magenta is characterized by ostiolate pycnidia that are larger than those of closely related other Trematosphaeria species. This species also differs from other species in having longer conidiophores and hyaline and variably shaped, elliptical, ovoid ellipsoidal to allantoid conidia. Phylogenetic analyses inferred from combined ITS, SSU, LSU, and RPB2 sequences indicated that T. magenta is the phylogenetic sister to T. pertusa, type species of Trematosphaeria. Detailed descriptions and illustrations of both new species are provided.




Similar content being viewed by others
References
Ahmed SA, Van De Sande WWJ, Stevens DA, Fahal A, Van Diepeningen AD, Menken SBJ, de Hoog GS (2014) Revision of agents of black-grain eumycetoma in the order Pleosporales. Persoonia 33:141–154. https://doi.org/10.3767/003158514X684744
Alidadi A, Kowsari M, Javan-Nikkhah M, Karami S, Ariyawansa HA, Salehi Jouzani GR (2019) Deniquelata quercina sp. nov.; a new endophyte species from Persian oak in Iran. Phytotaxa 405(4):187–194. https://doi.org/10.11646/phytotaxa.405.4.2
Bazdid Vahdati F, Saeidi Mehrvarz S, Naqinezhad AR, Shahi Shavvon R (2014) Floristic characteristics of the Hyrcanian submountain forests (case study: Ata-Kuh forest). Caspian J Environ Sci 12(2):169–183
Beyma FHV (1929) Mykologische untersuchungen. Verhan-delingen Koninklijke Nederlandse Akademie van Weten-schappen Afdeling Natuurkunde 26(2):1–29
Boise J (1985) New combinations in the Pleomassariaceae and the Massarinaceae. Mycotaxon 22:477–482
Cannon PF, Kirk PM (2007) Fungal families of the world. CABI Bioscience, Wallingford
Crous PW, Verkley GJ, Groenewald JZ, Samson RA (2009) Fungal biodiversity. CBS Laboratory Manual Series 1:1–269. Centraalbureau voor Schimmelcultures, Utrecht
Crous PW, Carnegie AJ, Wingfield MJ, Sharma R, Mughini G, Noordeloos ME, Santini A, Shouche YS, Bezerra JD, Dima B, Guarnaccia V (2019) Fungal planet description sheets: 868–950. Persoonia 42:291
Daniels J (1961) Chaetomium piluliferum sp. nov., the perfect state of Botryotrichum piluliferum. Trans Br Mycol Soc 44(1):79–IN7. https://doi.org/10.1016/S0007-1536(61)80009-0
Groenewald JZ, Nakashima C, Nishikawa J, Shin HD, Park JH, Jama AN, Groenewald M, Braun U, Crous PW (2013) Species concepts in Cercospora: spotting the weeds among the roses. Stud Mycol 75:115–170. https://doi.org/10.3114/sim0012
Hall TA (1999) BioEdit: a user–friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755. https://doi.org/10.1093/bioinformatics/17.8.754
Katoh K, Standley K (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Kushwaha RKS, Agrawal SC (1976) A new keratinophilic species of Botryotrichum. Trans Mycol Soc Japan.
Lee SB, Milgroom MG, Taylor JW (1988) A rapid, high yield mini-prep method for isolation of total genomic DNA from fungi. Fungal Genet Rep 35(1):23–24. https://doi.org/10.4148/19414765.1531
Liu D (2011) Molecular detection of human bacterial pathogens. CRC press, Boca Raton
Liu YJ, Whelen S, Hall BD (1999) Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit. Mol Biol Evol 16(12):1799–1808. https://doi.org/10.1093/oxfordjournals.molbev.a026092
Lumbsch HT, Huhndorf SM (2010) Myconet volume 14. Part one. Outline of Ascomycota—2009. Part two. Notes on Ascomycete systematics. Nos. 4751–5113. Fieldiana Life Earth Sci 2010(1):1–64
Maharachchikumbura SN, Hyde KD, Jones EBG, McKenzie EHC, Huang SK, Abdel-Wahab MA, Daranagama DA, Dayarathne M, D’souza MJ, Goonasekara ID, Hongsanan S, Jayawardena RS, Kirk PM, Konta S, Liu JK, Liu ZY, Norphanphoun C, Pang KL, Perera RH, Senanayake IC, Shang Q, Shenoy BD, Xiao Y, Bahkali AH, Kang J, Somrothipol S, Suetrong S, Wen T, Xu J (2015) Towards a natural classification and backbone tree for Sordariomycetes. Fungal Divers 72(1):199–301. https://doi.org/10.1007/s13225-015-0331-z
Maharachchikumbura SS, Hyde KD, Jones EG, McKenzie EHC, Bhat JD, Dayarathne MC, Huang SK, Norphanphoun C, Senanayake IC, Perera RH, Shang QJ (2016) Families of Sordariomycetes. Fungal Divers 79(1):1–317. https://doi.org/10.1007/s13225-016-0369-6
Nylander JA (2004) Bayesian phylogenetics and the evolution of gall wasps (Doctoral dissertation, Acta Universitatis Upsaliensis)
O'Donnell K, Cigelnik E (1997) Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol Phylogenet Evol 7:103–116. https://doi.org/10.1006/mpev.1996.0376
Rakesh K, Rai AN, Vyas KM (1994) A new species of Botryotrichum from Uttar Pradesh. Indian Phytopathol 47(2):181–182
Rannala B, Yang Z (1996) Probability distribution of molecular evolutionary trees: a new method of phylogenetic inference. J Mol Evol 43:304–31110. https://doi.org/10.1007/BF02338839
Rehner SA, Samuels GJ (1995) Molecular systematics of the Hypocreales: a teleomorph gene phylogeny and the status of their anamorphs. Can J Bot 73:816–823
Saccardo PA (1886) Sylloge Fungorum. Italy, Padua. 807 pp
Saccardo PA (1893) Mycetes aliquot australienses. Series quarta, Hedwigi 32:56–59
Schultes NP, Strzalkowski N, Li DW (2019) Botryotrichum domesticum sp. nov., a new hyphomycete from an indoor environment. Botany 97(6):311–319. https://doi.org/10.1139/cjb-2018-0196
Spegazzini C (1899) Fungi Argentini novi vel critici. Anales del Museo Nacional de Historia Natural Buenos Aires 6:81–365
Spegazzini C (1926) Contribucíon al conocimiento de la flora micológica de las Sierras de Córdoba. Boletín de la Academia Nacional de Ciencias en Córdoba 29(2–3):113–190
Subrahmanyam A (1979) Botryotrichum indicum A. Subrahm. sp. nov. Acta Mycologica 15(1):175–177. https://doi.org/10.5586/am.1979.010
Suetrong S, Hyde KD, Zhang Y, Bahkali AH, Jones EG (2011) Trematosphaeriaceae fam. nov. (Dothideomycetes, Ascomycota). Cryptogam Mycol 32(4):343–359. https://doi.org/10.7872/crym.v32.iss4.2011.343
Sung GH, Sung JM, Hywel-Jones NL, Spatafora JW (2007) A multi-gene phylogeny of Clavicipitaceae (Ascomycota, Fungi): identification of localized incongruence using a combinational bootstrap approach. Mol Phylogenet Evol 44(3):1204–1223. https://doi.org/10.1016/j.ympev.2007.03.011
Swofford DL (2002) Phylogenetic analysis using parsimony (* and other methods). b10
Tanaka K, Harada Y, Barr ME (2005) Trematosphaeria: taxonomic concepts, new species from Japan and key to species. Fungal Divers 19:145–156
Tanaka K, Hirayama K, Yonezawa H, Sato G, Toriyabe A, Kudo H, Tanaka K, Hirayama K, Yonezawa H, Sato G, Toriyabe A, Kudo H, Hashimoto A, Matsumura M, Harada Y, Kurihara Y, Shirouzu T (2015) Revision of the Massarineae (Pleosporales, Dothideomycetes). Stud Mycol 82:75–136. https://doi.org/10.1016/j.simyco.2015.10.002
Thambugala KM, Hyde KD, Tanaka K, Tian Q, Wanasinghe DN, Ariyawansa HA, Jayasiri SC, Boonmee S, Camporesi E, Hashimoto A, Hirayama K, Schumacher RK, Promputtha I, Liu ZY (2015) Towards a natural classification and backbone tree for Lophiostomataceae, Floricolaceae, and Amorosiaceae fam. nov. Fungal Divers 74:199–266
Waksman SA (1922) A method for counting the number of fungi in the soil. J Bacteriol 7(3):339
Wang XW, Houbraken J, Groenewald JZ, Meijer M, Andersen B, Nielsen KF, Crous PW, Samson RA (2016) Diversity and taxonomy of Chaetomium and chaetomium-like fungi from indoor environments. Stud Mycol 84:145–224. https://doi.org/10.1016/j.simyco.2016.11.005
Wang XW, Yang FY, Meijer M, Kraak B, Sun BD, Jiang YL, Wu YM, Bai FY, Seifert KA, Crous PW, Samson RA (2019) Redefining Humicola sensu stricto and related genera in the Chaetomiaceae. Stud Mycol 93:65–153. https://doi.org/10.1016/j.simyco.2018.07.001
White TJ, Bruns T, Lee SJWT, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocol 18:315–322
Winter G (1885) New North American fungi. J Mycolog, 1:101–102
Wu ZH, Wang TH, Huang W, Qu YB (2001) A simplified method for chromosome DNA preparation from filamentous Fungi. Mycosystema 20:575–577
Zhang Y, Fournier J, Pointing SB, Hyde KD (2008) Are Melanomma pulvis-pyrius and Trematosphaeria pertusa congeneric? Fungal Divers 33:47–60
Zhaxybayeva O, Gogarten JP (2002) Bootstrap, Bayesian probability and maximum likelihood mapping: exploring new tools for comparative genome analyses. BMC Genomics 3:4. https://doi.org/10.1186/1471-2164-3-4
Zhou Y, Gong G, Zhang S, Liu N, Wang J, Li P, Yu X (2014) A new species of the genus Trematosphaeria from China. Mycol Prog 13(1):33–43. https://doi.org/10.1007/s11557-013-0889-3
Acknowledgments
The authors would like to thank all staff of the Microbial Biotechnology Department, Agricultural Biotechnology Research Institute of Iran (ABRII), for their kind technical help.
Funding
This research was supported by a grant from Agricultural Biotechnology Research institute of Iran (ABRII), Agricultural Research, Education and Extension Organization (AREEO).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Amin Alidadi and Sepideh Akbari Vala. The first draft of the manuscript was written by Gholamreza Salehi Jouzani and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Data transparency
The authors declare that all data and materials as well as software application support our published claims and comply with field standards.
Additional information
Section Editor: Hans-Josef Schroers
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 531 kb)
Rights and permissions
About this article
Cite this article
Alidadi, A., Vala, S.A. & Jouzani, G.S. Botryotrichum iranicum sp. nov. and Trematosphaeria magenta sp. nov. as two new species from Iran. Mycol Progress 19, 1575–1586 (2020). https://doi.org/10.1007/s11557-020-01648-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-020-01648-7